Bilans d’évènement
Life Science Committee meeting with Danish Industry on MDR
Discussions emphasized that the future of the industry in Europe depends on decisions made today, especially in a global context shaped by the rising.
On Wednesday April 15, the Danish French Chamber of Commerce had the pleasure to host the Life Science Committee x Danish Industry in our HQ.
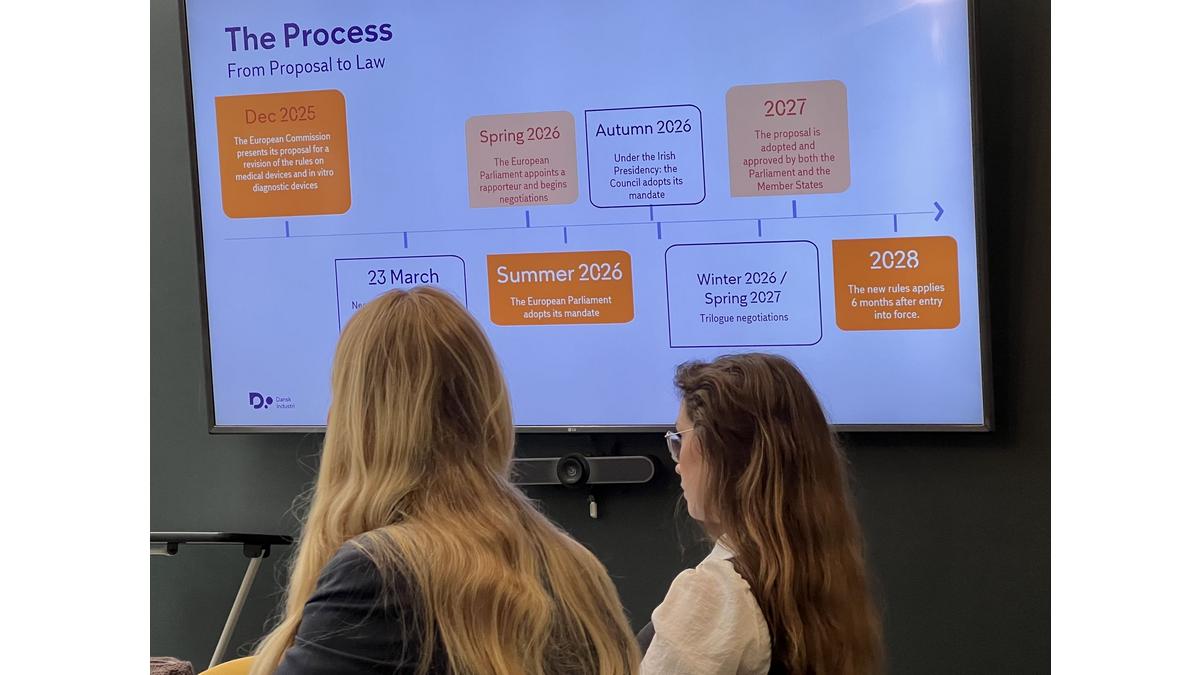
The meeting with Simone Harritshøj Sachmann, Senior Adviser in DI Life Science, highlighted how heavy bureaucracy and national regulatory fragmentation in the EU, especially under the Medical Device Regulation (MDR), are weakening the competitiveness of the EU Life Sciences industry.
Uncertainty, driven by complex rules, geopolitical tensions, and global competition shaped by the rising competitiveness of China and increasingly protectionist policies in the United States, was identified as main barriers to investment and growth.
While the European Commission is considering a shift toward less regulation and greater efficiency, concrete changes are unlikely short term. Participants stressed the urgent need for faster action to support innovation, improve patient access, and ensure Europe remains competitive globally.
Thank you to all participants for contributing to this enriching discussion.